ElectroChemistry 3
Previously , we have discussed all about Conductors and Electrolytes
Now , we'll be diving straight into the First main topic of Electrochemistry and that is
Electrolysis !
Electrolysis is defined as the process of decomposing ionic compounds into their respective elements by passing a direct electric current through the compound in a fluid form .
- What does that mean ?
When electricity is passed through an electrolytic solution , the ions move towards the electrodes according to the charges . That is , cations to cathode and anions to anode .
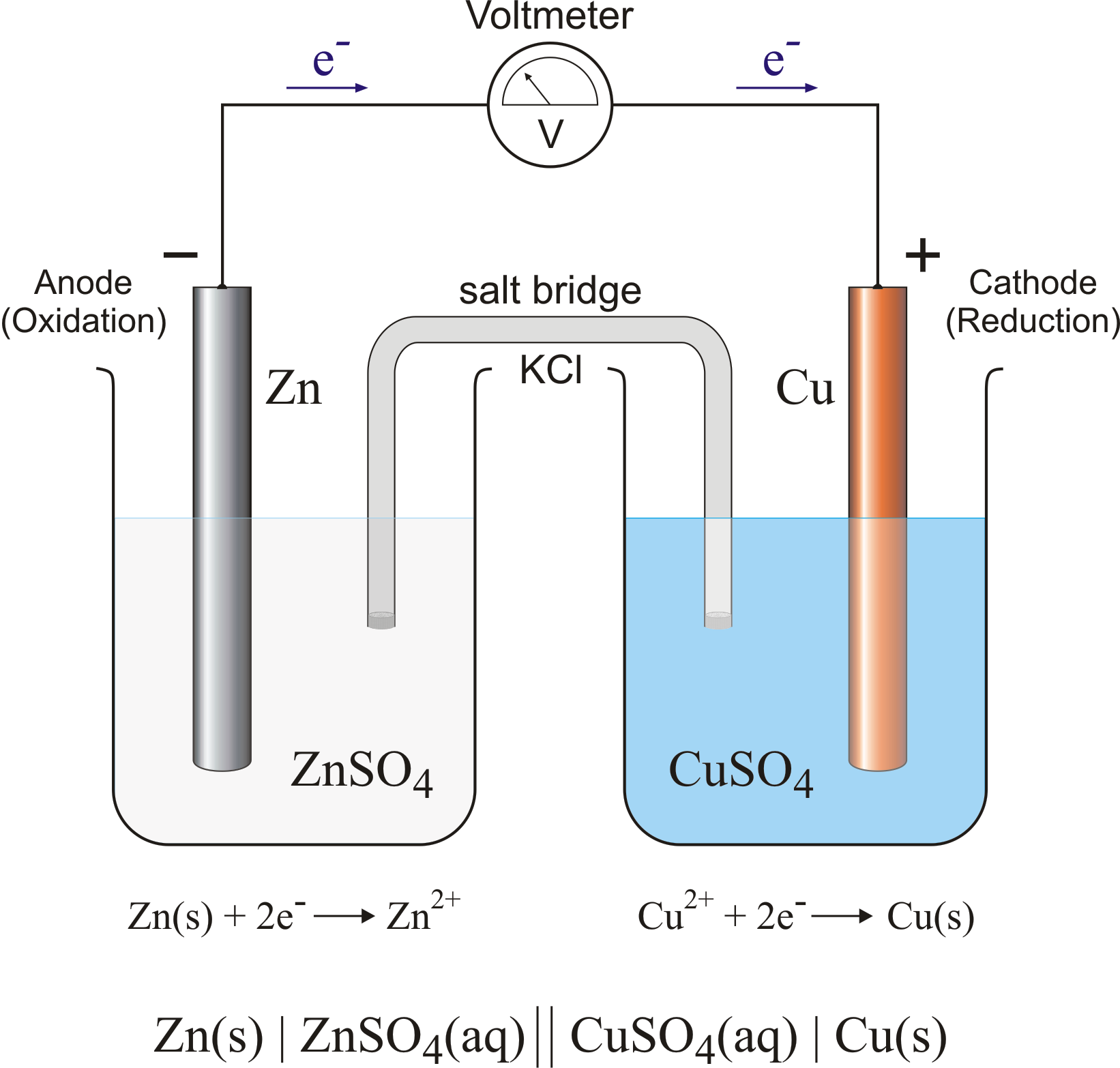
- Electrolysis is carried out in Electrolytic cell
- What is an Electrolytic cell ?
It is a setup in which there are two electrodes , anode and cathode respectively . Anode is connected to the negative terminal while cathode is connected to the positive terminal of the battery .
This is how an electrolytic cell looks . We'll discuss about some terms which are mentioned in the diagram above like Salt Bridge , and the different equations written soon
Oxidation and Reduction :
Oxidation : Oxidation is the loss of electrons during a reaction by a molecule, atom or ion .
Reduction : Reduction is the gain of electrons during a reaction by a molecule, atom or ion .
In an electrolytic cell ,
- At anode , Oxidation takes place
- At cathode , Reduction takes place
A small anagram to remember that is
anOx , redCat
where 'an-' is anode , 'Ox-' is oxidation , 'red-' is reduction and 'Cat-' is cathode .
Comments
Post a Comment